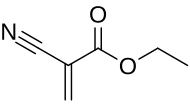
Ethyl cyanoacrylate
Names
Preferred IUPAC name
Ethyl 2-cyanoprop-2-enoate
Other names
Ethyl 2-cyanoacrylate; ECA; Ethyl alpha-cyanoacrylate; 910EM; ace-ee; CN2; CN4; Cemedine 3000rs; Krazy glue; KH502; Permabond 105 : Permabond 200; Super glue; Pro grip 4000; TK 200; TK 201; Cyanolite 201; Cyanacrine; Cyano-Veneer
Identifiers
ChEMBL
ChemSpider
ECHA InfoCard 100.027.628
EC Number
RTECS number
UNII
UN number
1993
InChI=1S/C6H7NO2/c1-3-9-6(8)5(2)4-7/h2-3H2,1H3
Y Key: FGBJXOREULPLGL-UHFFFAOYSA-N
Y InChI=1/C6H7NO2/c1-3-9-6(8)5(2)4-7/h2-3H2,1H3
Key: FGBJXOREULPLGL-UHFFFAOYAG
Properties
C 6 H 7 N O 2
Molar mass
−1
Density
1.06 g/mL
Melting point
−22 °C (−8 °F; 251 K)
Boiling point
54 to 56 °C (129 to 133 °F; 327 to 329 K) at 3 mmHg
Polymerises
Hazards
GHS labelling[ 1]
Warning
H315 , H319 , H335
P261 , P305+P351+P338
Flash point
83 °C (181 °F; 356 K)
0.2 ppm
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Ethyl cyanoacrylate (ECA ), a cyanoacrylate ester , is an ethyl ester of 2-cyano-acrylic acid . It is a colorless liquid with low viscosity and a faint sweet smell in pure form. It is the main component of cyanoacrylate glues and can be encountered under many trade names .[ 2] acetone , methyl ethyl ketone , nitromethane , and methylene chloride .[ 3] polymerizes rapidly in presence of moisture .
Production
Ethyl cyanoacrylate is prepared by the condensation of formaldehyde with ethyl cyanoacetate :
NCCH2 CO2 C2 H5 + CH2 O → H2 C=C(CN)CO2 C2 H5 + H2 O This exothermic reaction affords the polymer, which is subsequently sintered , thermally "cracked" to give the monomer . Alternatively, it can be prepared by the ethoxycarbonylation of cyanoacetylene .[ 2]
Applications
Ethyl cyanoacrylate is used for gluing.
In forensics, cyanoacrylate ester has excellent non-destructive impressioning abilities, which are especially important when lifting fingerprints from delicate evidence items, or when the prints could not be lifted using traditional means such as fingerprinting powder. The procedure involves heating the acrylate in a sealed chamber. Its fumes then react with deposited proteins that form into a white, stable, and clear print outlines. The resulting prints could be used 'as is' or enhanced further by staining them with darker pigments.[ 4] [ 5] Liquid bandage systems use the less toxic n -butyloctyl cyanoacrylates .
Safety
In the U.S., the threshold limit value for ECA is 0.2 ppm. It is a strong irritant to the lungs and eyes .
See also
References
^ GHS: Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
^ a b Ohara, Takashi; Sato, Takahisa; Shimizu, Noboru; Prescher, Günter; Schwind, Helmut; Weiberg, Otto; Marten, Klaus; Greim, Helmut; Shaffer, Timothy D.; Nandi, Partha (2020). "Acrylic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry 1– 21. doi :10.1002/14356007.a01_161.pub4 . ISBN 9783527303854 ^ "Cyanoacrylate Technical Data" . palmlabsadhesives.com . Archived from the original on 3 June 2009. Retrieved 12 January 2022 .^ Bumbrah, Gurvinder Singh (2017). "Cyanoacrylate fuming method for detection of latent fingermarks: a review" . Egyptian Journal of Forensic Sciences . 7 (1): 4. doi :10.1186/s41935-017-0009-7 PMC 5514188 PMID 28781896 . ^ Mutter, Nicole; Deacon, Paul; Farrugia, Kevin J. (2018-11-30). "The effect of cyanoacrylate fuming on subsequent protein stain enhancement of fingermarks in blood". Journal of Forensic Identification . 68 (4): 545– 556. S2CID 104335657 .